
Straub, T., Grimaud, C., Gilfillan, G.D., Mitterweger, A. A sequence motif within chromatin entry sites directs MSL establishment on the Drosophila X chromosome. 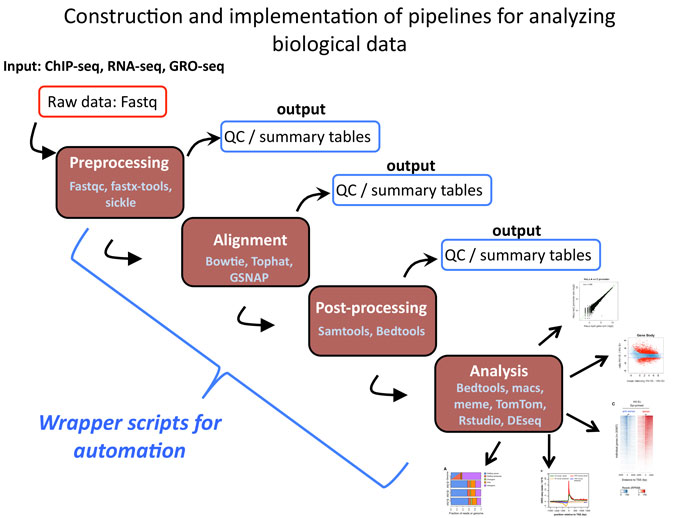 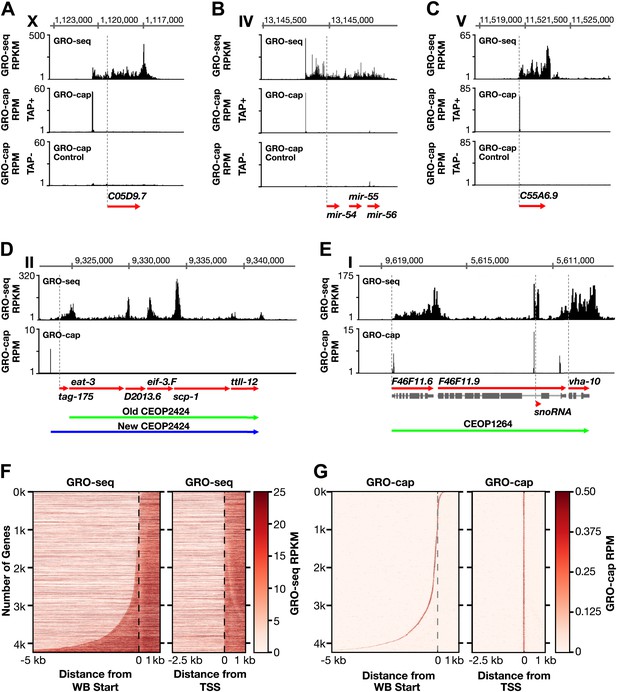
Drosophila dosage compensation: a complex voyage to the X chromosome. Probabilistic modeling of Hi-C contact maps eliminates systematic biases to characterize global chromosomal architecture. Altered nuclear retention of mRNAs containing inverted repeats in human embryonic stem cells: functional role of a nuclear noncoding RNA. RNA-RNA interactions enable specific targeting of noncoding RNAs to nascent Pre-mRNAs and chromatin sites. Unambiguous identification of miRNA:target site interactions by different types of ligation reactions. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Super-enhancers in the control of cell identity and disease. A high-resolution map of the three-dimensional chromatin interactome in human cells. Topological domains in mammalian genomes identified by analysis of chromatin interactions. Chromatin connectivity maps reveal dynamic promoter–enhancer long-range associations. Extensive promoter-centered chromatin interactions provide a topological basis for transcription regulation. An oestrogen-receptor-alpha-bound human chromatin interactome. A 3D map of the human genome at kilobase resolution reveals principles of chromatin looping. Comprehensive mapping of long-range interactions reveals folding principles of the human genome. The Xist lncRNA exploits three-dimensional genome architecture to spread across the X chromosome. The genomic binding sites of a noncoding RNA. Genomic maps of long noncoding RNA occupancy reveal principles of RNA–chromatin interactions. The long noncoding RNAs NEAT1 and MALAT1 bind active chromatin sites. Genome regulation by long noncoding RNAs. Non-coding RNA: a new frontier in regulatory biology. Landscape of transcription in human cells. We would appreciate it if you could operate the website and give us feedbacks to improve it and continue notify us about new data sets via the google form.Djebali, S. If you have such a dataset please don’t hesitate to fill in the google form and we will contact you back. We are crowd-sourcing additional datasets that may not be available in the public domain but are available within laboratories. We have summarized all of the curated datasets in this google form. An ER and breast cancer-centered database for dissecting the biological and technical diversity and variation of estrogen receptor-relevant NGS experiments and the confound ER regulomes in breast cancer. A browser supporting statistical cutoff-based gene list query function to export genes regulated by E2 under users’ defined contexts. A browser allowing single gene-based visualization of E2-induced expressional changes and ER proximal binding at users’ selected genes of interest. A uniformly processed and crowd-sourced multi-omic database with detailed experimental documentation summary. We have curated 136 published NGS data sets from 2004-2022 across 19 breast cancer cell lines and generated a browser for simplified queries. It aims to document and integrate the majority of publicly available estrogen-stimulated next generation sequencing data sets (including RNA-seq, microarray, ChIP-seq, ATAC-seq, ChIA-PET, Hi-C, GRO-seq, etc), and establish a comprehensive database to allow users’ customized data mining and visualization. We are pleased to introduce the EstroGene Project -a comprehensive multi-omic NGS database focusing on estrogen receptor function in breast cancer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
